2020/11/06(金)
Kazunori Yoshikawa, Chief of Executive Officer, Veneno Technologies
Innovative drug discovery targeting ion channels with the molecular evolution of disulfide-rich peptides
Kazunori Yoshikawa, Chief of Executive Officer, Veneno Technologies

[Summary]
● Business definition and contribution to society and technology
Membrane proteins, such as ion channels and transporters, are known to be important but difficult targets for drug discovery. Veneno Technologies Co. Ltd. has developed a simple and rapid screening method for DRP (Disulfide-Rich Peptide), which has multiple disulfide bonds in the molecule and has recently been attracting attention as a platform for drug discovery, using evolutionary molecular engineering techniques PERISSTM. Furthermore, we have established a low-cost mass production method using E. coli to address the production of DRPs, which is a major issue in DRP drug discovery due to their complex structure.
Utilizing these core technologies, we will initially pursue the exploration of novel DRPs targeting ion channels, where DRPs have been shown to be useful. By providing a complete solution from DRP discovery to manufacturing and supply, the company aims to create new drug discovery opportunities based on DRPs, accelerate research and development in the pharmaceutical industry, and contribute to the creation of new drugs.
-Why target the ion channel and how the business contributes to society
Ion channels are transmembrane proteins that have been strongly implicated in various diseases such as cancer, immune disorders, and cardiovascular diseases. Although many pharmaceutical companies have pursued research and development of new drugs targeting ion channels mainly using small molecules, most drug discovery projects have been terminated due to poor channel selectivity of candidate compound. Therefore ion channel modulators are still limited to classical drugs such as nifedipine and phenytoin etc. in the WHO essential list, and many other similar drugs.
We approach these historically important but difficult ion channel targets with a cutting edge technology PERISSTM that enables DRP-based drug discovery, opening up the possibility of drug discovery that no one has ever been able to achieve before. Our technology will make many ion channels potential targets for drug discovery, enabling numerous pharmaceutical companies to approach the development of therapeutics for diseases involving ion channels.
-Why DRP as a Modality and Characteristics of PERISSTM and Mass Synthesis
A large number of DRPs that act strongly on ion channels have been found in nature and from biotoxins, and due to their excellent in vivo stability and low immunogenicity derived from their unique molecular structure, they are attracting a great deal of attention as next-generation drug discovery modalities that target membrane protein including ion channels. Several pharmaceutical companies are using DRPs as template peptides for drug discovery research and development, but the lack of manufacturing methods and efficient screening methods for DRPs with their complex structures is a major hurdle in DRP drug discovery.
Our PERISSTM is an evolutionary molecularly engineered DRP screening method that utilizes the periplasmic space of E. coli to enable high-throughput screening from our DRP-focused library. Our DRP-focused library consists of plasmid DNA of unprecedented size that encodes one billion DRP variants. However, the size is not the only advantage. Because our library has been constructed using a DRP as template that have evolved in nature to target ion channels and neural receptors, it is possible to generate more active and more selective molecules with a much higher probability than conventional random sequence libraries.
The PERISSTM method uses the nature of plasmid incompatibility, that is one E. coli acts as one reaction field, and one DRP variant translated from one plasmid can be screened as an affinity for the target membrane protein. This innovative method enables screening of one billion size libraries using only E. coli culture equipment without the need for expensive HTS equipment, and is capable of finding candidate DRPs from libraries in a short period time (about six months). On the other hand, existing technologies such as the phage display method and mRNA display based on cell-free translation systems are well known. However, these are not suitable for screening for DRPs due to their inability to correctly generate DRP molecules and need for ad hoc preparation of target molecule such as ion channel in the evaluation system.
In addition, DRPs have been synthesized mainly by peptide chemical synthesis, but it is difficult to synthesize such a complex higher-order structure in a sufficient quantity and requires high production costs. In collaboration with a Japanese chemical company, we have developed a technology for secreting DRP from the culture supernatant of E. coli, aiming at simple and low-cost mass production of DRP. By providing solutions to the two major challenges of screening and manufacturing, we can lower the barriers to research and development and dramatically increase the speed of development.
● Marketability
Nearly all global pharma companies are included as our target customers (pipeline buyer and users of drug discovery partnering), since they have identified many diseases involving ion channels as targets for development. The global market for conventional small molecule drugs targeting ion channels is reported to be approximately $12billion per year, but there are a number of ion channels that have yet to be explored due to the difficulties of drug discovery. DRPs can open up new markets and the size of the pharmaceutical market in the future is immense.
In recent years, the number of peptide drugs approved by the U.S. FDA has grown to rival protein and antibody drugs in terms of the number of new drugs approved, and the peptide drug market is expected to grow to $23 billion by 2020. Each of our pipeline products has the potential to become blockbusters (annual sales of more than $1 billion) in the markets and is an attractive market.
● Proposer/team biographies/awards received
Tadashi Kimura, Ph.D. the inventor of Veneno Technologies, won the Special Jury Prize in NEDO's Technology commercialization program 2015 and the Grand Prize in Global Tech EDGE NEXT (2019) at the University of Tsukuba. He has also been selected for the JST Research Results Development Program (START), a program to create new industries from universities. In July this year, Veneno Technologies Co. Ltd. was incorporated with Kazunori Yoshikawa, who has extensive experience in bio-venture management, as President and CEO.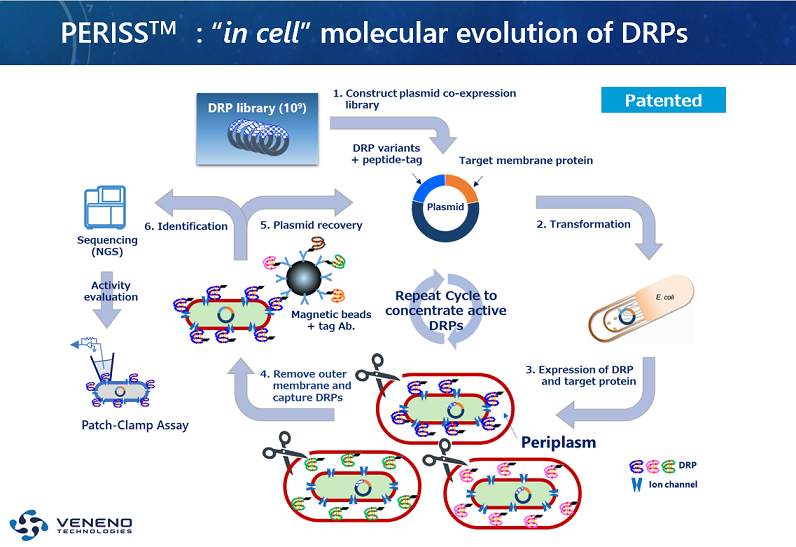
Figure: Conceptual diagram of the PERISSTM method
In the PERISSTM cycle, membrane proteins such as ion channels, which are target molecules, are expressed in the inner membrane and each DRP variant in the periplasmic space simultaneously, and then DRPs that interact with the target molecules are recovered together with genetic information. By repeating this cycle, peptides that bind specifically to the target molecule are enriched, and the target DRP candidates can be screened from the DRP-focused library in a short period of time.
[Profile]
Over 30 years of experiences in the pharmaceutical industry. Well-experienced in non-clinical and clinical development of multiple global products, including recombinant proteins and antibody drugs. Later, as a management member of several biotech ventures, involved in starting company, raising capital, forming business alliances with pharmaceutical companies.